Description
Kit for microbiome collection and DNA/RNA preservation of oral, skin, fecal, rectal, vaginal, and soil microbiome samples, & transport/storage at ambient temperatures – all in the same tube.
Kit contents: 1 feces catcher, 2 swabs, 1 barcode-labeled iSWAB-MB vial (1ml iSWAB-MB preservation solution), 1 plastic “biohazard” bag with absorbent pad, 1 extra barcode label, 1 “Exempt Human Specimen” label, and a box which may be used to mail back the sample
Please let us know the required kit quantity to receive a price quote.
• iSWAB-Microbiome maintains the status quo at the time of collection
• Provides a representative snapshot of the microbial community that remains unchanged from collection to processing of oral, gut/fecal, skin, vaginal, and soil samples
• Microbial presentation maintained intact from time of collection for weeks at room temperature
• DNA and RNA from collected microorganisms remains stable at room temperature for up to 40 days for DNA, and 21 days for RNA
• Non-toxic stabilization; no organic, hazardous solvent fixatives or detergents.
• The stabilization buffer efficiently inactivates bacteria, fungi, spores, and viruses allowing safe transport of biological sample
• No bloom effect correction needed for microbiome data. Both aerobic and anaerobic bacterial communities stabilized from collection to processing.
• Ambient temperature transport and storage with no cold chain involvement.
• Swab-free sample transport: Improved sample integrity and decreased sample processing time
• Traceable: LIMS compatible unique barcodes included for each iSWAB tube enable efficient traceability and storage
• iSWAB-MB buffer allows the isolation of microbial DNA or RNA with minimal or no human genomic DNA contamination.
• Purified microbial DNA or RNA isolated from collected samples are compatible with NGS, microarrays, qPCR, and PCR.
• Scalable and customizable platform – please contact us when requiring high capacities (of up to 20g of fecal or soil material)
iSWAB-Microbiome – A snapshot of the microbiome from point of microbiome collection to processing
:Microbial content and diversity within collected gut, rectal, vaginal, skin, oral, or soil samples can provide a wealth of clues about human and animal health. However, current microbiome collection methods subject the samples to harsh and stressful stabilization techniques such as freezing or harsh organic solvents. Both stabilization approaches have a high probability of altering the microbial representation in the sample, which could lead to misleading results and hinder proper modeling or data analysis.
iSWAB-Microbiome (MB) overcomes these hurdles through collection at room temperature applying a non-toxic stabilizing technology which maintains the status quo at the time of collection, whilst bacteria, fungi, spores, and viruses are inactivated allowing safe handling of the collected sample.
Devices only for collecting microbial/viral DNA and RNA from oral, nasal, soil, skin, and fecal samples. Microbiome analysis (from Fecal, skin, vaginal, soil, etc.) qPCR, microarray, and NGS for microbiome research in health, wellness, and forensics.
Product Description
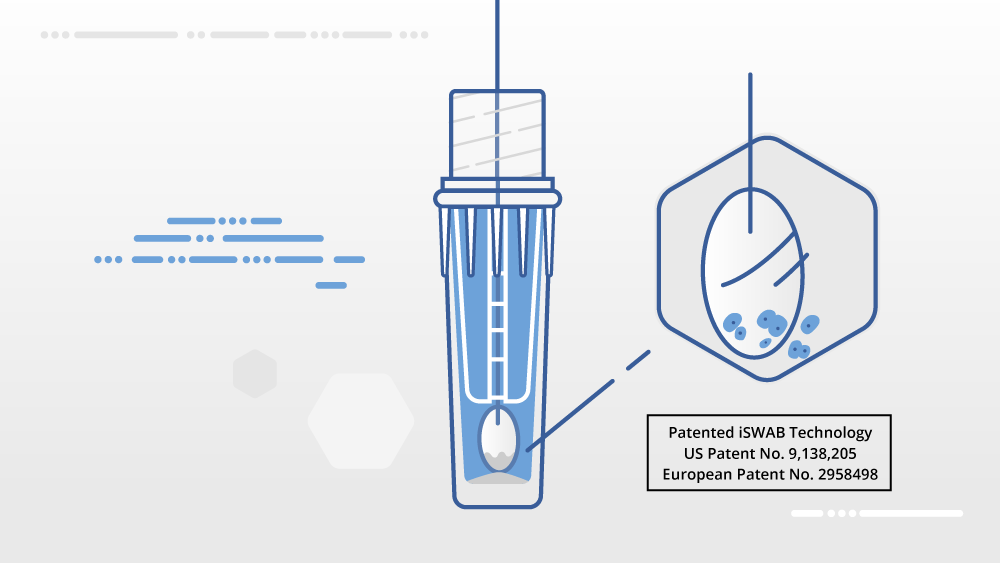
The iSWAB™-Microbiome (iSWAB™-MB) collection device consists of a collection device that is pre-filled with 1.0 mL of the iSWAB™-MB non-toxic, stabilizing buffer and fitted with a proprietary insert. The insert is designed to optimize the release of specimens collected with swabs into the stabilizing buffer, creating a minimal footprint and allowing for swab-free transport of specimens. The iSWAB™-MB buffer is designed to maintain the status quo of samples at the time of collection, providing a representative snapshot of the microbial community that remains unchanged from the point of collection to processing.
Intended Use
iSWAB™-Microbiome collection device is a non-invasive, non-toxic sample collection technology intended for stable ambient transport and storage of microbial/viral DNA and RNA from various sample types including fecal, vaginal, skin, soil, nasal, oral, and saliva. Purified microbial/viral DNA or RNA isolated from collected samples are compatible with qPCR, microarray, and NGS for microbiome research in health, wellness, and forensics.
Indication for Use
The iSWAB™-Microbiome sample collection technology is intended for specimen collection of biological samples. It is a non-invasive, non-toxic sample collection method and is intended to be used for long-term room temperature, stable transport, and storage of microbial/viral DNA and RNA from various sample types including fecal, vaginal, skin, soil, nasal, and oral. The iSWAB-Microbiome device is designed to recover and stabilize collected samples from a nylon flocked or molded swab head and then the swab is discarded at the point of collection. For saliva collection, only a spit funnel is needed to facilitate the collection of saliva into the iSWAB™-Microbiome device
Use for SARS-CoV-2 Samples
The iSWAB™-Microbiome device is ideal for stabilization and inactivation of upper and lower respiratory human specimens supsected of containing SARS-CoV-2.
Storage and Stability
Sample stability and storage, post-collection: 21 days for microbial RNA, 40 days for microbial DNA, and 42 days for SARS-Cov-2 samples at 15-30° C. If longer storage time is required, collected samples can be stored at -20 to -80° C.
Downstream Processing:
Bacterial genomic DNA may be purified with the QIAmp DNA Microbiome kit,Thermo´s MagMAX Microbiome Ultra Nucleic Acid Isolation Kit or PureLink Microbiome DNA Purification kit, Macherey-Nagel´s NucleoMag or Nucleospin DNA Microbiome Kit, or Qiagen´s QIAamp® DNA Mini kit for further analysis such as 16S rRNA NGS, metagenomics NGS, qPCR, PCR etc.
Bacterial RNA may be purified with Thermo´s MagMAX Microbiome Ultra Nucleic Acid Isolation Kit, or Qiagen´s RNeasy mini kit for any further analysis such as RNA-Seq or microarrays for microbial transcriptomics, qRT-PCR, RT-PCR etc.
Viral RNA extraction kits from Omega Biotek, Qiagen, Thermo Fisher, Magbio, Beckman Coulter, and Perkin Elmer have been validated for viral RNA extraction from iSWAB-MB collection tubes
.See also MAWI DNA Technologies Extraction Methods Overview for further information.